-
MCI pt interested in drugs to prevent/delay AD
No drugs/supplements shown to improve MCI cognitive sx or delay progression, per AAN;1 EFNS2 concurs that none have proven efficacy for preventing/ delaying AD - Cholinesterase inhibitors. Given lack of evidence in MCI studies,2 clinicians may choose not to offer these, per AAN; if using off-label for MCI, discuss lack of evidence w/ pt in advance1
- Other drugs/supplements. No evidence shows vitamin E, ginkgo, or anti‐inflammatory drugs to be helpful, per EFNS;2 no evidence for statins or aspirin in preventing cognitive decline/dementia; no additional vitamins or supplements have shown benefit3
- Actions that might prevent/delay dementia:3 SBP ≤130 mmHg for ages ≥40 yo; hearing aid if warranted; limited EtOH (<21 units/wk); smoking cessation/avoidance, ↓2nd-hand tobacco smoke/air pollution exposure; ↓obesity/T2DM; avoid traumatic brain injury, per Lancet Commission
- Trials. Clinical trials may be of interest (eg, Alzheimer's Assn TrialMatch, ClinicalTrials.gov)1
Footnotes 1 AAN 2018. Petersen RC, et al. Practice Guideline Update Summary: Mild Cognitive Impairment. Neurology. 2018. Jan 16;90(3):126–135. PubMed article
2 EFNS 2010. Hort J, et al. EFNS Guidelines for the Diagnosis and Management of Alzheimer’s Disease. Eur J Neurol. 2010. Oct;17(10):1236–1248. PubMed abstract
3 Lancet Commission 2020. Livingston G, et al. Dementia Prevention, Intervention, and Care: 2020 Report of the Lancet Commission. Lancet. 2020. Aug 8;396(10248):413-446. Full-text article
These 12 modifiable factors might prevent/delay up to 40% of dementia cases:
- • HTN (strongest evidence for effect on dementia)
- • EtOH. Heavy drinking linked w/ brain changes, cognitive impairment, dementia. Drinking >21 units/wk & long-term abstinence both linked w/ 17% dementia increase vs <14 units/wk.
- • DM. T2DM is a risk factor, but unclear whether any specific drug improves risk; intensive DM control doesn’t lower dementia risk.
- • Obesity
- • Traumatic brain injury (concussion, skull fracture, edema, brain injury/bleed)
- • Air pollution
- • Education. Less ed in early life (eg, <45 yo) affects cognitive reserve, whereas midlife (45–65 yo) & later-life ed influences reserve, triggering of neuropathological developments.
- • Hearing impairment
- • Smoking. Cessation ameliorates risk, even in later life. Passive smoking is a less considered modifiable risk factor for dementia.
- • Depression may be a risk or an effect of dementia.
- • Physical inactivity
- • Infrequent social contact. Evidence on social interventions is limited.
Though evidence insufficient to support specific activities, physical, cognitive, & social activity all recommended; promote secondary ed to ↑cognitive reserve (though ed <20 yo may be most important). Support general health via optimal sleep, nutrition, etc, as pts in good physical health can sustain greater neuropathological burden. Depression & sleep disturbance may be risks &/or effects of dementia. Mediterranean/Scandinavian diet might help those w/ intact cognition prevent cognitive decline, as part of healthy lifestyle.
-
AD pt awaiting decision on AD-targeted pharmaco-tx
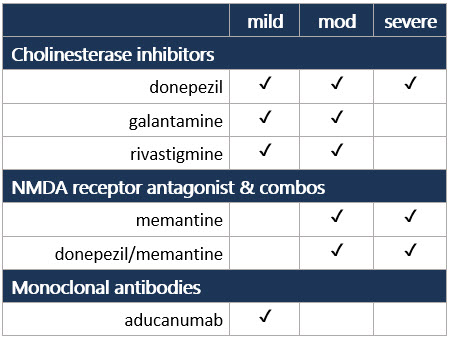
Individualize decision to trial cholinesterase inhibitor or memantine (weak recommendation per ACP).1 Although drugs recommended by some groups,2-6 cognition benefits aren’t clinically meaningful on average1 & are of uncertain clinical importance7,8 FDA approval by AD stage9 - Factor drug benefit/harm, tolerability, adverse effects, ease, cost; insufficient evidence to compare effectiveness of options1-3
- Cholinesterase inhibitor. Most evidence is in mild-to-mod9 AD.5 EFNS recommends considering @ dx for all AD severities;3 CCC recommends for most AD pts at all stages, also for AD pts w/ cerebrovascular component2
- Combo cholinesterase inhibitor + add-on memantine. CCC concluded no clear benefit to combo;2 others concur evidence lacking.1,7 However, EFNS concluded combo’s desirable effects outweigh undesirable ones for mod-to-severe AD (weak recommendation).4 Memantine not well studied in mild9 AD1
- Aducanumab reduces amyloid plaques on amyloid PET, but improvements in function, cognition, QOL, maintenance of independence, or survival not demonstrated; adverse events common (ARIA-E in 41%) & may ↓cognition, QOL, independence, and survival; cost of tx is high11
- Drug trial endpoint/duration. Evidence on duration insufficient.1,3 Periodically assess cognitive benefit & adverse drug effects.8 Benefits (eg, improved or stabilized), if any, generally seen w/in 3mo.1 If desired effects (eg, cognition stabilization) not seen w/in ~12wk, d/c drug, per AGS8
- Discontinuation. If slowing decline no longer a goal (eg, poor QOL in advanced AD), don’t continue.1 CCC suggests d/c'ing cholinesterase inhibitors based on pt/caregiver risk/benefit appraisal (incl risk/futility from comorbidities), intolerable adverse effects, non-adherence, rate of decline greater on tx than pre-tx, etc.2 If d/c'ing, CCC suggests tapering. If d/c'ing due to lack of effectiveness, monitor x1-3mo post-d/c; if observable decline, consider reinstating tx2
Not recommended - Don’t use as AD tx: vitamin B or E, multi-component nutrition supplement (unless deficiency); PUFA;10 aspirin3
- Insufficient/conflicting evidence for AD: Ginkgo biloba, NSAIDs, statins, estrogens, selegiline, nicergoline, vinpocetine, piracetam, pentoxyphylins, Cerebrolysin (pig brain extract), per EFNS.3 Evidence for cognitive training insufficient6
Footnotes 1 ACP 2008. Qaseem A, et al. Current Pharmacologic Treatment of Dementia: A Clinical Practice Guideline From the American College of Physicians and the American Academy of Family Physicians. Ann Intern Med. 2008. Mar 4;148(5):370-8. PubMed abstract
Cholinesterase inhibitors & memantine tx
- • Balance harms vs benefits, which may be modest to none.
- • Benefits are statistically significant, yet aren’t clinically significant for cognition on average; benefits are modest for global assessments (incl ADL, QOL, etc). Limited evidence is suggestive (not conclusive) that an unknown proportion of pts have clinically important responses. It's unclear how to predict such pts, so evidence doesn't support prescribing for every dementia pt.
2 CCC 2014. Moore A, et al. Fourth Canadian Consensus Conference on the Diagnosis and Treatment of Dementia. Can Fam Physician. 2014. May;60(5):433-438. PubMed article
3 EFNS 2010. Hort J, et al. EFNS Guidelines for the Diagnosis and Management of Alzheimer's Disease. Eur J Neurol. 2010. Oct;17:1236-1248. PubMed abstract
4 EFNS‐ENS/EAN 2015. Schmidt R, et al. EFNS‐ENS /EAN Guideline on Concomitant Use of Cholinesterase Inhibitors and Memantine in Moderate to Severe Alzheimer's Disease. Eur J Neurol. 2015. Jun;22(6):889-98. PubMed abstract
5 WHO 2015. Cholinesterase Inhibitors and Memantine for Treatment of Dementia. Dementia. Evidence-Based Recommendations for Management of Dementia in Non-Specialized Health Settings. Updated 2015. Accessed online 10/14/22
- • Cholinesterase inhibitor use recommended to be focused on mild-to-mod AD, d/t evidence.
- • Memantine may be considered for mod-to-severe AD (conditional recommendation, very low evidence).
6 Lancet Commission 2020. Livingston G, et al. Dementia Prevention, Intervention, and Care: 2020 Report of the Lancet Commission. Lancet. 2020. Aug 8;396(10248):413-446. Full-text article
- • Mild-to-mod dz. Cholinesterase inhibitors have modest role for cognition/ADL improvement.
- • Mod & severe dz. Memantine can be used alone or in combination.
7 Fink H, et al. Benefits and Harms of Prescription Drugs and Supplements for Treatment of Clinical Alzheimer-Type Dementia: A Systematic Review and Meta-analysis. Ann Intern Med. 2020. May 19;172(10):656-668. PubMed abstract
Though data show slight ↓cognitive decline short-term (cholinesterase inhibitors, memantine) & slight ↓functional decline (cholinesterase inhibitors)—differences vs placebo are of uncertain clinical importance.
Cholinesterase inhibitors. Overall across AD stages: Compared w/ placebo, cholinesterase inhibitors linked to small cognition improvement on average, 0-to-small functional improvement, no difference in moderate-or-better global clinical impression (evidence strength: mostly low).
Memantine combo. For mod-to-severe AD pts on cholinesterase inhibitors: Compared w/ placebo, add-on memantine inconsistently improved cognition, improved global clinical impression, didn't improve fxn (evidence strength: low to insufficient).
8 AGS 2015. American Geriatrics Society. Choosing Wisely. Revised April 23, 2015. Accessed online 5/25/23
Unclear whether RCT cognitive test results are clinically meaningful; studies haven't clarified long-term risk/benefit.
9 Alzheimer stage by scoring system or symptoms
MMSE
- • Alzheimer's Assn:* 24-20 mild, 20-13 mod, <12 severe
- • Other:† 26-20 mild, 19-10 mod, <10 severe
CDR‡
- • CDR (per domain or global): 1 mild, 2 mod, 3 severe
-
- • CDR-SOB dementia stage: 4.5-9 mild, 9.5-15.5 mod, 16-18 severe
Symptoms§
Mild. Trouble making sense of world, realizes something's wrong. Memory loss, poor judgment/bad decisions, ↓spontaneity/initiative, slow @ ADL, repeats questions, trouble handling money/bills, wanders/lost, loses/misplaces items. Mood/personality change, ↑anxiety/aggression
Moderate. More intensive supervision & care required; worse memory loss & confusion; unable to learn; language/number difficulties; difficulty w/ logic & thought organization. Short attention span, problems coping w/ new situations, difficulty w/ multi-step tasks (eg, dressing), problem recognizing friends/family, hallucination/delusion/paranoia, impulsive/inappropriate action/speech/anger outburst, repetitive speech/movement, occasional muscle twitch. Restless, agitated, anxious, tearful, wandering (esp late afternoon/evening)
Severe. Unable to communicate; completely dependent on others for care; may be bedridden. Difficulty swallowing, wt loss, seizures, skin infxn, groans/moans/grunts, ↑sleeping, bowel/bladder control loss
* Medical Tests. Alzheimer's Association. Accessed online 7/3/20
† Forchetti CM. Treating Patients With Moderate to Severe Alzheimer's Disease. Prim Care Comp J Clin Psy. 2005. 7(4):155–161. PubMed article
‡ O'Bryant SE, et al. Staging Dementia Using Clinical Dementia Rating Scale Sum of Boxes Scores: A Texas Alzheimer's Research Consortium Study. Arch Neurol. 2008. Aug;65(8):1091-5. PubMed abstract
§ What Are the Signs of Alzheimer's Disease? National Institute on Aging. May 16, 2017. Accessed online 6/17/20
10 WHO 2019. Risk Reduction of Cognitive Decline and Dementia: WHO Guidelines. World Health Organization. Feb. 5, 2019. Accessed online 6/17/20
11AAN 2022. Day GS, et al. Aducanumab Use in Symptomatic Alzheimer Disease. Evidence in Focus. A Report of the AAN Guidelines Subcommittee. Neurology. 2022. Apr 12;98(15):619-631. Neurology article
-
AD pt awaiting decision on tx behavioral/psych sx (agitation, psychosis, delirium, etc)
Explore triggers/exacerbators (environment, pain, constipation, infxn, drug effect, depression, etc).1 Try non-drug 1st2-4 (education, exercise, sensory tx, music, etc).1 EFNS notes non-cognitive benefits of cholinesterase inhibitors, memantine;1 others conclude no good evidence for neuropsych sx5,6 For depression: Use non-drug tx 1st if sx non-severe - Mild-to-mod depression in dementia: WHO recommends offering cognitive behavioral tx, interpersonal tx, structured counseling, etc; antidepressants aren’t 1st line3
- Major depressive disorder/severe depression or if non-drug tx fails: Consider antidepressant trial;5 use SSRIs, not TCAs,3 which may have adverse anticholinergic effects1
- For apathy: A review found a small benefit for methylphenidate on 1 (but not all) apathy scales4
For agitation/psychosis/aggression: Use non-drug tx 1st, d/t significant risks w/ antipsychotics4 - Antipsychotics: Use only if sx severe, dangerous, or significantly distressing to pt, after discussing risk/benefit, per APA;2 use only if unresponsive to non-drug tx & cholinesterase inhibitors, per EFNS.1 WHO prefers specialist input when using antipsychotics & if possible, informed pt consent3
- Drug options. Start low, titrate to min effective dose.2 Consider aripiprazole, olanzapine, risperidone1 for severe agitation, aggression, psychosis w/ risk of harm to pt/others. Carbamazepine may help aggression1
- Don't use thioridazine, chlorpromazine, or trazodone for behavioral/psych sx, per WHO;3 (CCC considers role of trazodone, quetiapine, SSRIs uncertain for agitation5). If no delirium, don’t use haloperidol 1st line.2 Don’t use valproate for agitation/aggression.5 Trazodone, BZDs ↑fall risk.4 Don’t use long-acting injectable antipsychotic (unless concurrent chronic psychotic dz)2
- If (+) response, discuss risk/benefit of possible tapering; attempt taper & w/d w/in 4mo of tx start (unless sx recurrence w/ tapering). Monitor at least qmo during taper plus ≥4mo post-d/c2
- If (-) clinically significant response after 4-wk trial at adequate dose, taper & w/d. Monitor at least qmo during taper plus ≥4mo post-d/c2
- If significant adverse effect, review risk/benefit to determine if taper or d/c is appropriate. Monitor at least qmo during taper plus ≥4mo post-d/c2
For delirium, address risks/exacerbators. Use non-drug tx; antipsychotics, BZDs linked to ↑morbidity/mortality4 - Risks: polypharmacy/predisposing meds, pain, dehydration, illness (eg, UTI, fecal impaction), sensory impairment, environment change
- Inpt: Multidisciplinary intervention reduces delirium, incl fluid repletion, feeding support, mobilization/therapeutic activity, vision/hearing adaptation, sleep enhancement (not sleep meds), infxn prevention; managing pain, constipation, hypoxia, etc
- Ineffective: antipsychotics, cholinesterase inhibitors, BZDs aren’t efficacious for delirium. No evidence for melatonin
Footnotes 1 EFNS 2010. Hort J, et al. EFNS Guidelines for the Diagnosis and Management of Alzheimer’s Disease. Eur J Neurol. 2010. Oct;17(10):1236–1248. PubMed abstract
Behavioral & psychiatric sx:
- • Cholinesterase inhibitors. Non-cognitive benefits have been reported, incl in mild-mod-severe AD, although uncertain which non-cognitive sx are most responsive (psychosis, apathy have been consistently reported). 1 RCT found donepezil didn’t improve agitation in mod-to-severe AD.
- • Memantine. Non-cognitive benefits seen in an analysis of studies in pts w/ MMSE <20: delusion, agitation/aggression, irritability were most responsive.
- • Other drugs. 2nd-gen antipsychotics have fewer side effects; particular effects shown w/ risperidone for agitation/aggression & psychosis. Carbamazepine may help aggression. SSRIs, which don’t have the anticholinergic effects older TCAs do, may help depression.
2 APA 2016. The American Psychiatric Association Practice Guideline on the Use of Antipsychotics to Treat Agitation or Psychosis in Patients With Dementia. Am J Psychiatry. 2016. May 1;173:543. PubMed abstract
3 WHO 2015. Dementia. Evidence-Based Recommendations for Management of Dementia in Non-Specialized Health Settings. Updated 2015. Accessed online 10/14/22
4 Lancet Commission 2020. Livingston G, et al. Dementia Prevention, Intervention, and Care: 2020 Report of the Lancet Commission. Lancet. 2020. Aug 8;396(10248):413-446. Full-text article
5 CCC 2014. Moore A, et al. Fourth Canadian Consensus Conference on the Diagnosis and Treatment of Dementia. Can Fam Physician. 2014. May; 60(5):433-438. PubMed article
6 Fink H, et al. Benefits and Harms of Prescription Drugs and Supplements for Treatment of Clinical Alzheimer-Type Dementia: A Systematic Review and Meta-analysis. Ann Intern Med. 2020. May 19;172(10):656-668. PubMed abstract
Behavioral/psychological drugs. Prescriptions, supplements: evidence largely insufficient.
|