-
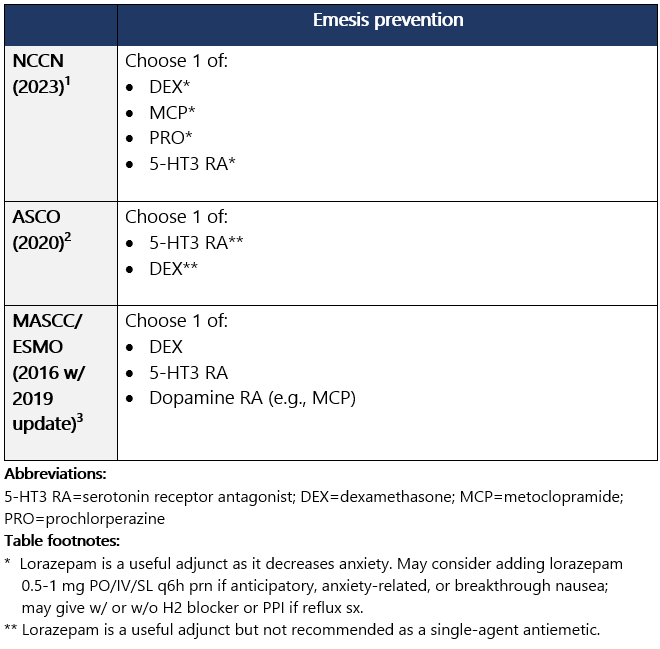
Candidate for prevention of N/V
TABLE | Emetic risk (%) by drug To determine emetic risk of parenteral chemo drug, tap here. Footnotes 1 NCCN 2023. National Comprehensive Cancer Network. Antiemesis (Version 1.2024). December 13, 2023. Accessed December 20, 2023
2 ASCO 2020. Hesketh PJ, et al. Antiemetics: ASCO Guideline Update. J Clin Oncol. 2020. Aug 20;38(24):2782-2797. Accessed October 19, 2020
3 MASCC/ESMO 2016/2019. Roila F, et al. 2016 MASCC and ESMO Guideline Update for the Prevention of Chemotherapy- and Radiotherapy-Induced Nausea and Vomiting and of Nausea and Vomiting in Advanced Cancer Patients. Ann Oncol. 2016. Sep;27(suppl 5):v119-v133. PDF
Aapro M, et al. MASCC/ESMO Antiemetic Guideline 2016 With Updates in 2019. June 2019. Multinational Association of Supportive Care in Cancer.
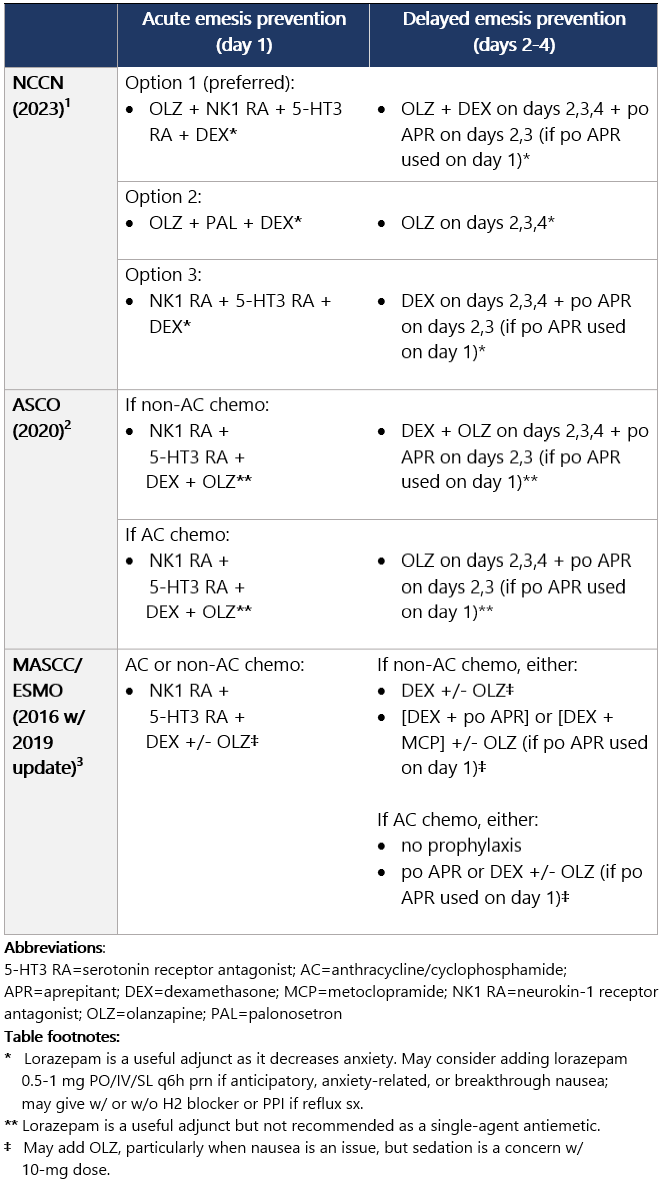
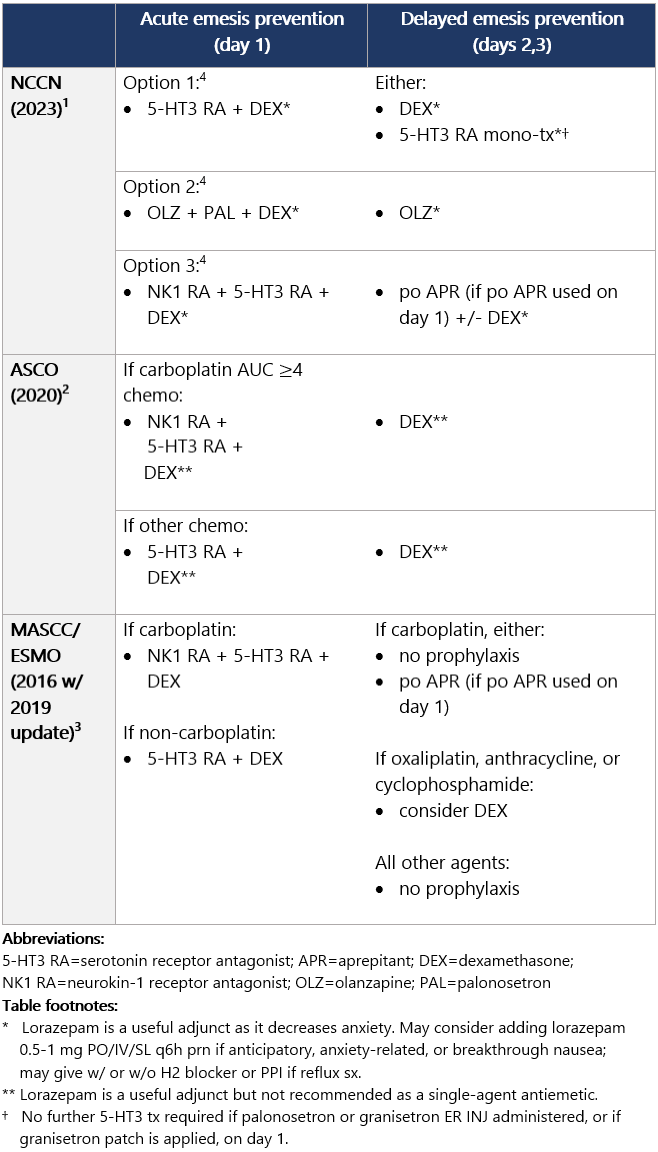
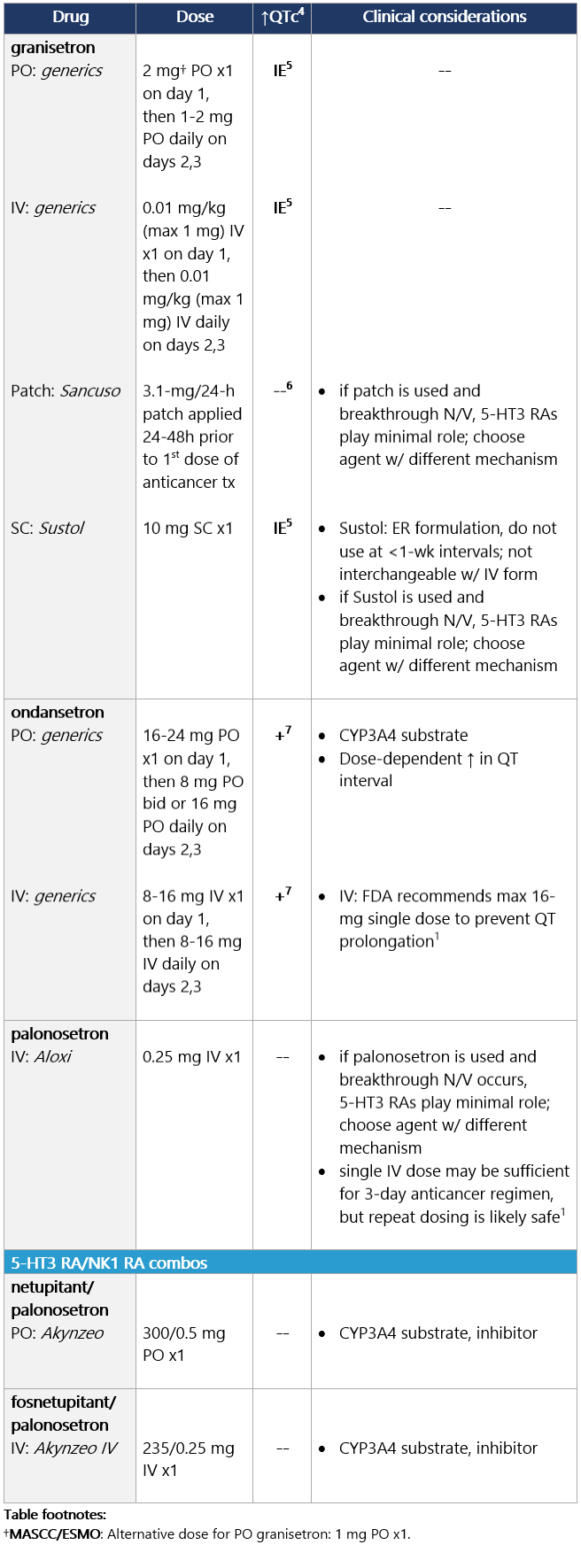
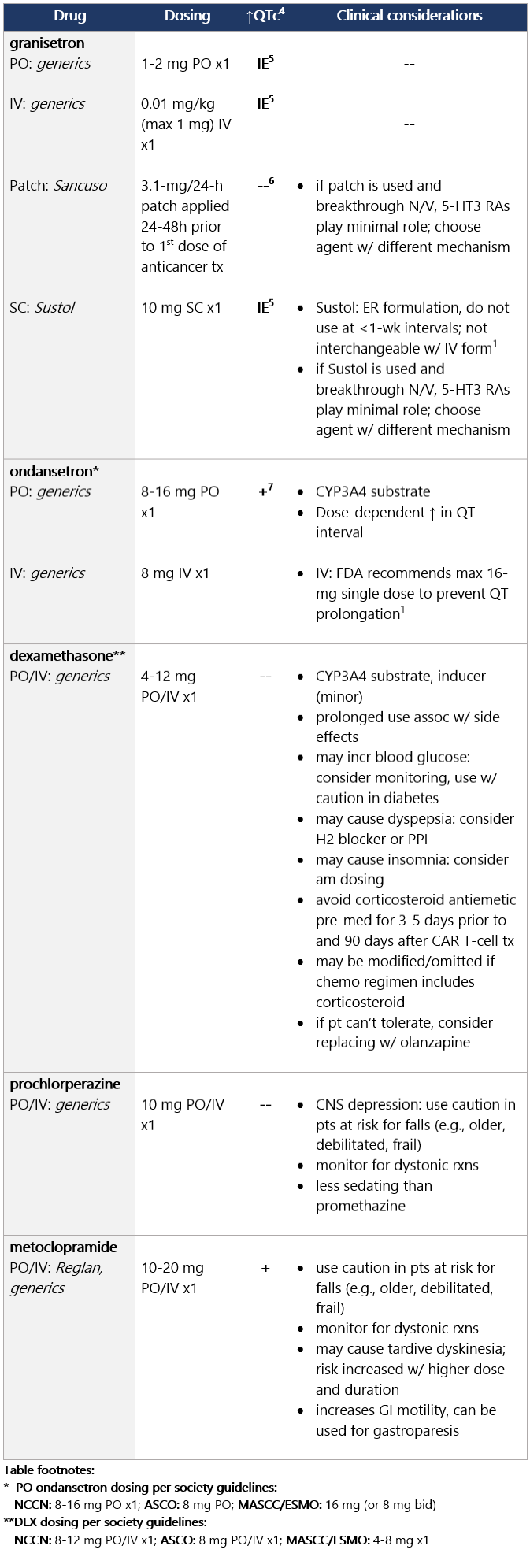
High (>90%) emetic risk drug Guideline recs for highly emetogenic parenteral chemo (for dosing, see drug tables below): 5-HT3 receptor antagonists Drug class considerations for 5-HT3 RAs: - Administer prior to 1st (and subsequent) dose of chemo. Administer 5-HT3 RAs regularly vs. prn.1
- 5-HT3 RAs are nonsedating; most common side effects are headache, constipation; educate pts on constipation mgmt.1
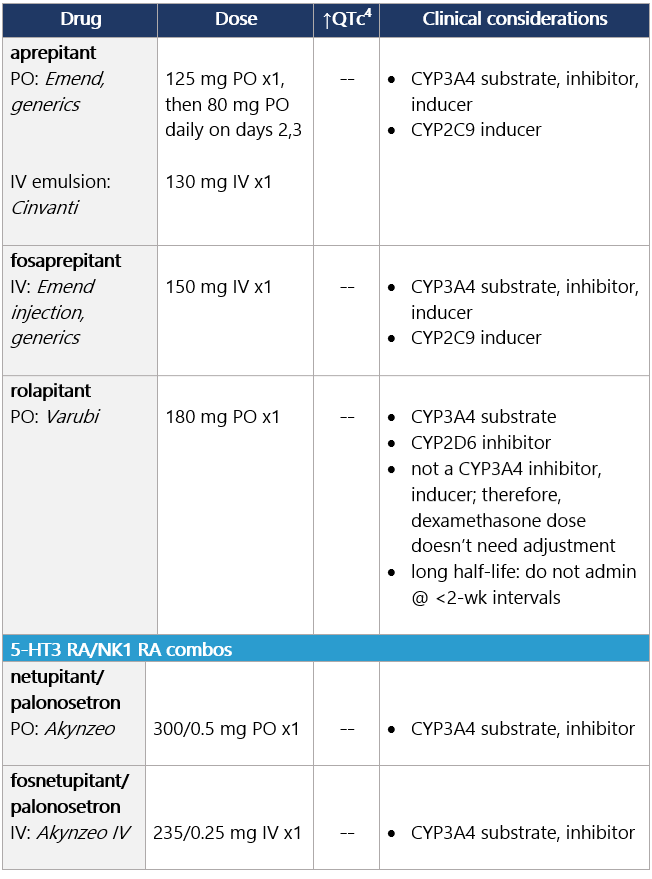
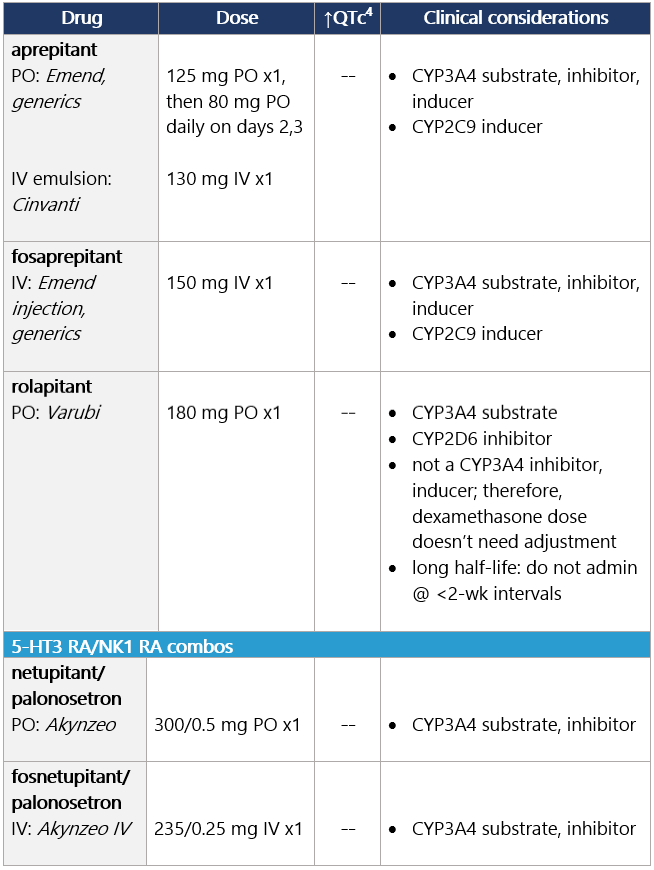
View epocrates drug info: Drug class considerations for NK1 RAs: - Place in therapy is for prevention, not tx, of CINV (e.g., breakthrough emesis); largest benefit in delayed CINV.
- Aprepitant (PO and INJ emulsion), fosaprepitant, netupitant, and fosnetupitant inhibit metabolism of DEX; rolapitant does not share this interaction.
- Data from two phase II studies support multiday dosing of netupitant/palonosetron in association w/ BEAM/FEAM conditioning regimens prior to hematopoietic cell transplant. An open-label RCT also supports multiday dosing of fosaprepitant. Studies on repeat dosing of aprepitant IV emulsion, fosnetupitant, and rolapitant are not available.
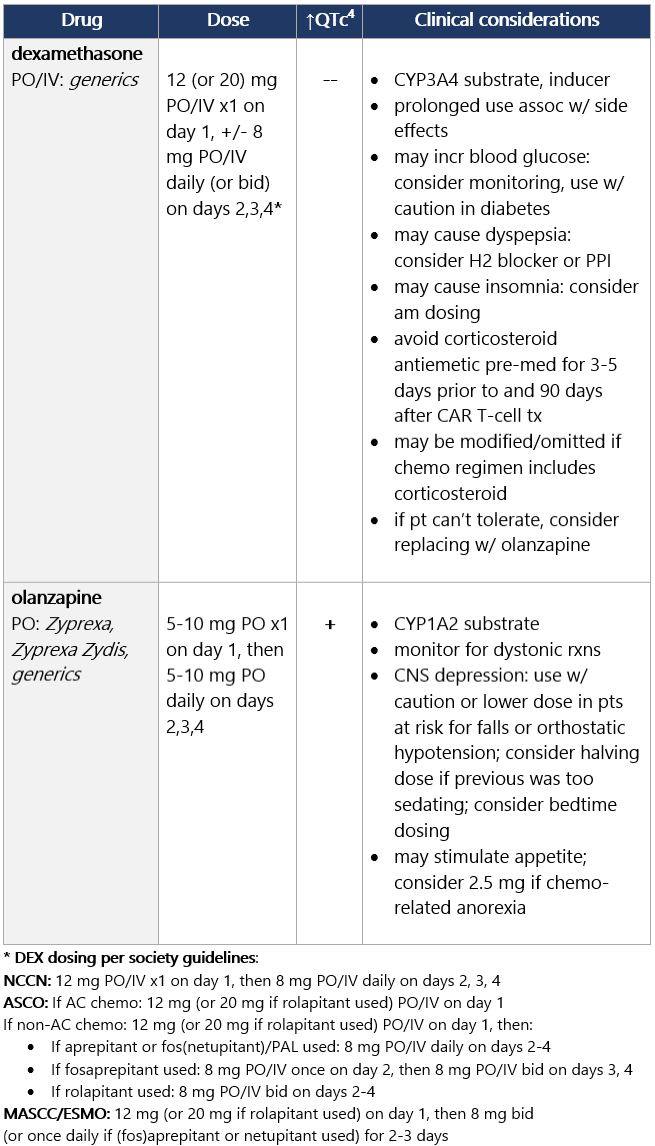
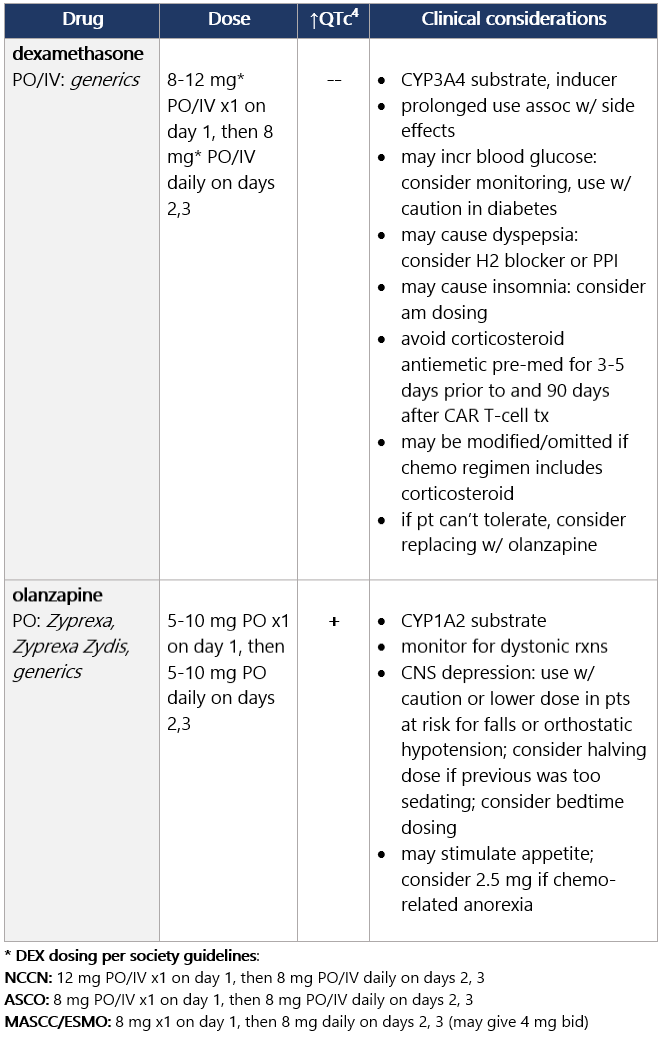
View epocrates drug info: Dexamethasone and olanzapine View epocrates drug info: Footnotes 1 NCCN 2023. National Comprehensive Cancer Network. Antiemesis (Version 1.2024). December 13, 2023. Accessed December 20, 2023
2 ASCO 2020. Hesketh PJ, et al. Antiemetics: ASCO Guideline Update. J Clin Oncol. 2020. 2020. Aug 20;38(24):2782-2797. Accessed October 19, 2020
• For adults, addition of a checkpoint inhibitor (CPI) to chemo doesn’t change the guideline rec for antiemetic regimen based on emetogenicity of chemo agents; CPIs are minimally emetogenic and don’t require a prophylactic antiemetic.
3 MASCC/ESMO 2016/2019. Roila F, et al. 2016 MASCC and ESMO Guideline Update for the Prevention of Chemotherapy- and Radiotherapy-Induced Nausea and Vomiting and of Nausea and Vomiting in Advanced Cancer Patients. Ann Oncol. 2016. Sep;27(suppl 5):v119-v133. PDF
Aapro M, et al. MASCC/ESMO Antiemetic Guideline 2016 With Updates in 2019. June 2019. Multinational Association of Supportive Care in Cancer.
4 Barni S, et al. Cardiotoxicity of Antiemetic Drugs in Oncology: An Overview of the Current State of the Art. Crit Rev Oncol Hematol. 2016. June;102:125-34. PubMed® abstract
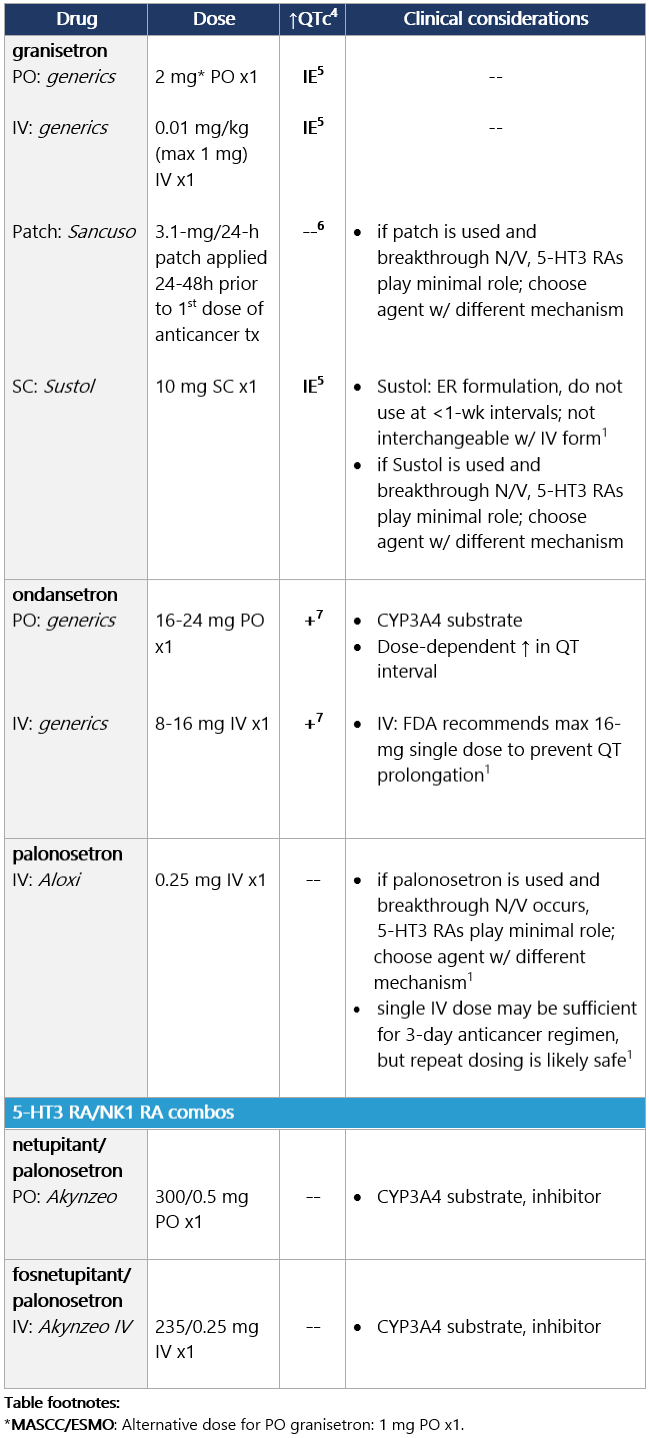
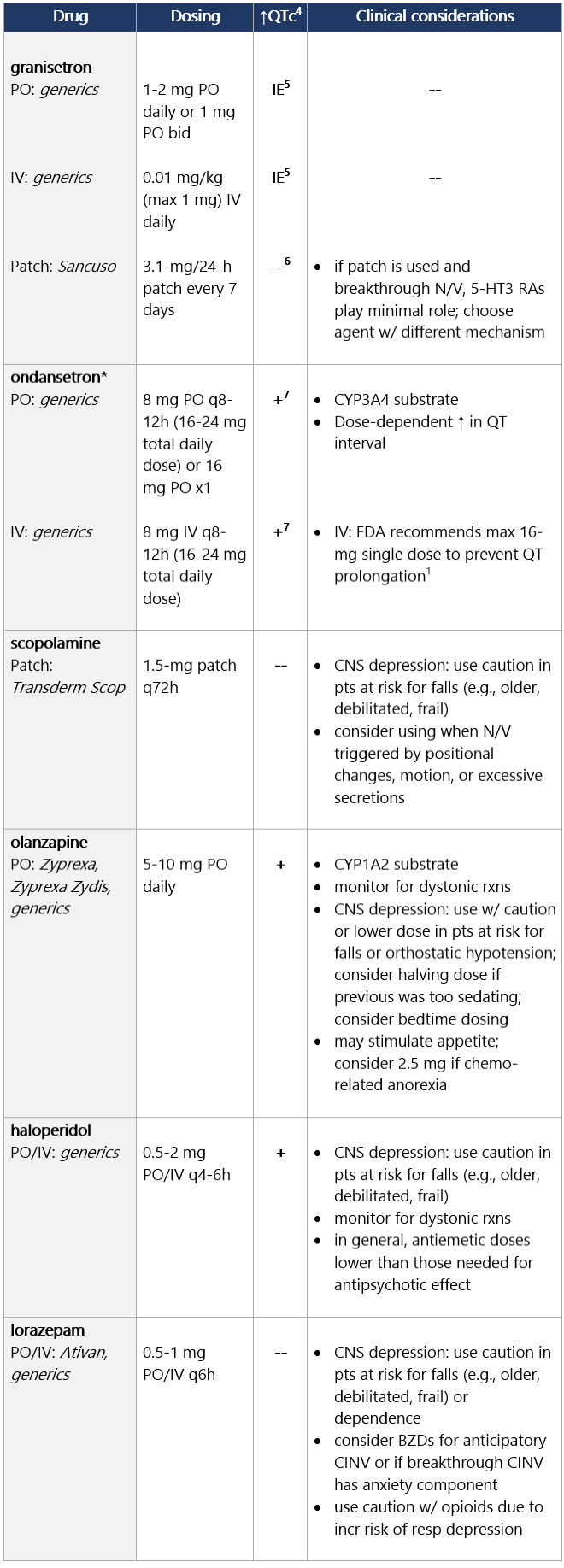
5 IE: insufficient evidence. QT prolongation data for PO/IV/SC granisetron are limited and inconsistent.
6 Granisetron transdermal doesn’t alter QT interval.
7 Ondansetron: dose-dependent QT change. Moderate (>30%-90%) emetic risk drug Guideline recs for moderately emetogenic parenteral chemo (for dosing, see drug tables below): 5-HT3 receptor antagonists Drug class considerations for 5-HT3 RAs: - Administer prior to 1st (and subsequent) dose of chemo. Administer 5-HT3 RAs regularly vs. prn.1
- 5-HT3 RAs are nonsedating; most common side effects are headache, constipation; educate pts on constipation mgmt.1
View epocrates drug info: Drug class considerations for NK1 RAs: - Place in therapy is for prevention, not tx, of CINV (e.g., breakthrough emesis); largest benefit in delayed CINV.
- Aprepitant (PO and INJ emulsion), fosaprepitant, netupitant, and fosnetupitant inhibit metabolism of DEX; rolapitant does not share this interaction.
- Data from two phase II studies support multiday dosing of netupitant/palonosetron in association w/ BEAM/FEAM conditioning regimens prior to hematopoietic cell transplant. An open-label RCT also supports multiday dosing of fosaprepitant. Studies on repeat dosing of aprepitant IV emulsion, fosnetupitant, and rolapitant are not available.
View epocrates drug info: Dexamethasone and olanzapine View epocrates drug info: Footnotes 1 NCCN 2023. National Comprehensive Cancer Network. Antiemesis (Version 1.2024). December 13, 2023. Accessed December 20, 2023
2 ASCO 2020. Hesketh PJ, et al. Antiemetics: ASCO Guideline Update. J Clin Oncol. 2020. 2020. Aug 20;38(24):2782-2797. Accessed October 19, 2020
• For adults, addition of a checkpoint inhibitor (CPI) to chemo doesn’t change the guideline rec for antiemetic regimen based on emetogenicity of chemo agents; CPIs are minimally emetogenic and don’t require a prophylactic antiemetic.
3 MASCC/ESMO 2016/2019. Roila F, et al. 2016 MASCC and ESMO Guideline Update for the Prevention of Chemotherapy- and Radiotherapy-Induced Nausea and Vomiting and of Nausea and Vomiting in Advanced Cancer Patients. Ann Oncol. 2016. Sep;27(suppl 5):v119-v133. PDF
Aapro M, et al. MASCC/ESMO Antiemetic Guideline 2016 With Updates in 2019. June 2019. Multinational Association of Supportive Care in Cancer.
4 Barni S, et al. Cardiotoxicity of Antiemetic Drugs in Oncology: An Overview of the Current State of the Art. Crit Rev Oncol Hematol. 2016. June;102:125-34. PubMed® abstract
5 IE: insufficient evidence. QT prolongation data for PO/IV/SC granisetron are limited and inconsistent.
6 Granisetron transdermal doesn’t alter QT interval.
7 Ondansetron: dose-dependent QT change. Low (10%-30%) or minimal (<10%) emetic risk drug If minimal emetic risk, no routine prophylaxis recommended. Guideline recs for low-emetogenic parenteral chemo (for dosing, see drug tables below): Start before anticancer tx; repeat daily for multiday regimens. 5-HT3 RAs, Corticosteroids, Phenothiazine, & Prokinetic View epocrates drug info: Footnotes 1 NCCN 2023. National Comprehensive Cancer Network. Antiemesis (Version 1.2024). December 13, 2023. Accessed December 20, 2023
2 ASCO 2020. Hesketh PJ, et al. Antiemetics: ASCO Guideline Update. J Clin Oncol. 2020. 2020. Aug 20;38(24):2782-2797. Accessed October 19, 2020
• For adults, addition of a checkpoint inhibitor (CPI) to chemo doesn’t change the guideline rec for antiemetic regimen based on emetogenicity of chemo agents; CPIs are minimally emetogenic and don’t require a prophylactic antiemetic.
3 MASCC/ESMO 2016/2019. Roila F, et al. 2016 MASCC and ESMO Guideline Update for the Prevention of Chemotherapy- and Radiotherapy-Induced Nausea and Vomiting and of Nausea and Vomiting in Advanced Cancer Patients. Ann Oncol. 2016. Sep;27(suppl 5):v119-v133. PDF
Aapro M, et al. MASCC/ESMO Antiemetic Guideline 2016 With Updates in 2019. June 2019. Multinational Association of Supportive Care in Cancer.
4 Barni S, et al. Cardiotoxicity of Antiemetic Drugs in Oncology: An Overview of the Current State of the Art. Crit Rev Oncol Hematol. 2016. June;102:125-34. PubMed® abstract
5 IE: insufficient evidence. QT prolongation data for PO/IV/SC granisetron are limited and inconsistent.
6 Granisetron transdermal doesn’t alter QT interval.
7 Ondansetron: dose-dependent QT change. TABLE | Emetic risk (%) by drug To determine emetic risk of oral chemo drug, tap here. Footnotes 1 NCCN 2023. National Comprehensive Cancer Network. Antiemesis (Version 1.2024). December 13, 2023. Accessed December 20, 2023
2 ASCO 2020. Hesketh PJ, et al. Antiemetics: ASCO Guideline Update. J Clin Oncol. 2020. 2020. Aug 20;38(24):2782-2797. Accessed October 19, 2020
• For adults, addition of a checkpoint inhibitor (CPI) to chemo doesn’t change the guideline rec for antiemetic regimen based on emetogenicity of chemo agents; CPIs are minimally emetogenic and don’t require a prophylactic antiemetic.
3 MASCC/ESMO 2016/2019. Roila F, et al. 2016 MASCC and ESMO Guideline Update for the Prevention of Chemotherapy- and Radiotherapy-Induced Nausea and Vomiting and of Nausea and Vomiting in Advanced Cancer Patients. Ann Oncol. 2016. Sep;27(suppl 5):v119-v133. PDF
Aapro M, et al. MASCC/ESMO Antiemetic Guideline 2016 With Updates in 2019. June 2019. Multinational Association of Supportive Care in Cancer.
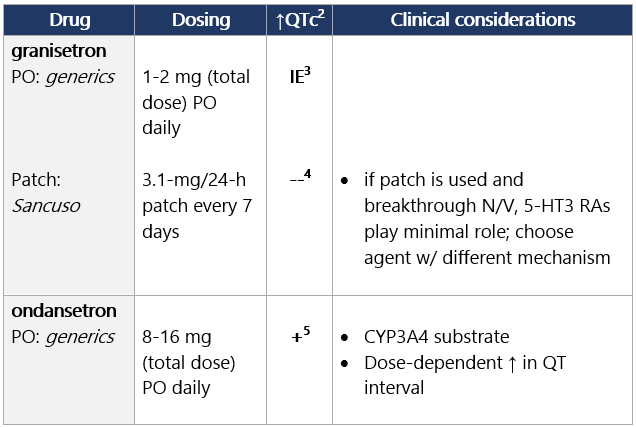
Moderate (>30%-90%) to high (≥90%) emetic risk Start 5-HT3 RA before anticancer tx and continue daily.1 5-HT3 receptor antagonists (in alpha order) View epocrates drug info: Footnotes 1 NCCN 2023. National Comprehensive Cancer Network. Antiemesis (Version 1.2024). December 13, 2023. Accessed December 20, 2023
2 Barni S, et al. Cardiotoxicity of Antiemetic Drugs in Oncology: An Overview of the Current State of the Art. Crit Rev Oncol Hematol. 2016. June;102:125-34. PubMed® abstract
3 IE: insufficient evidence. QT prolongation data for PO/IV/SC granisetron are limited and inconsistent.
4 Granisetron transdermal doesn’t alter QT interval.
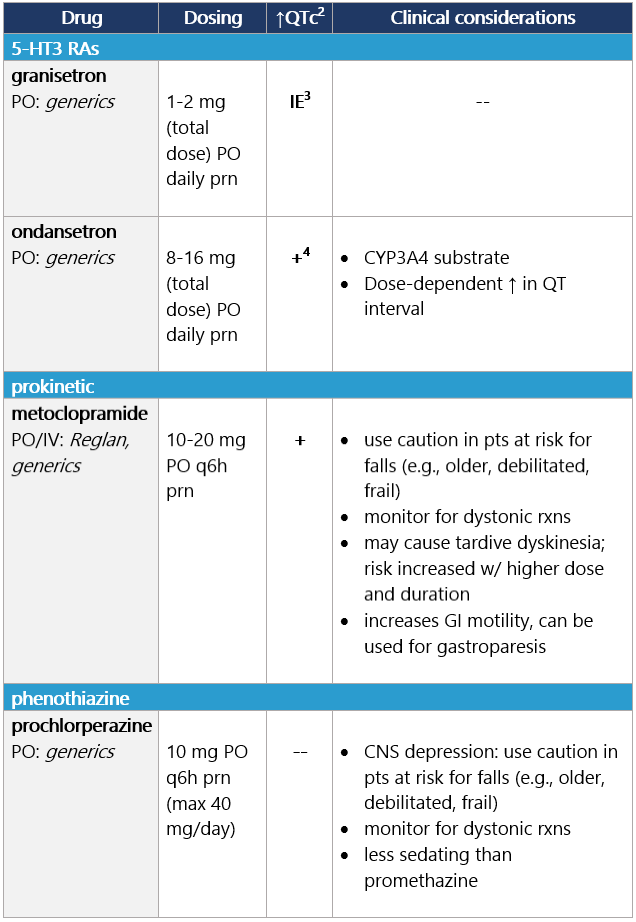
5 Ondansetron: dose-dependent QT change. Low (10%-30%) or minimal (<10%) emetic risk Start antiemetic before anticancer tx; continue daily prn.1 View epocrates drug info: Footnotes 1 NCCN 2023. National Comprehensive Cancer Network. Antiemesis (Version 1.2024). December 13, 2023. Accessed December 20, 2023
2 Barni S, et al. Cardiotoxicity of Antiemetic Drugs in Oncology: An Overview of the Current State of the Art. Crit Rev Oncol Hematol. 2016. June;102:125-34. PubMed® abstract
3 IE: insufficient evidence. QT prolongation data for PO/IV/SC granisetron are limited and inconsistent.
4 Ondansetron: dose-dependent QT change. -
Experiencing breakthrough N/V
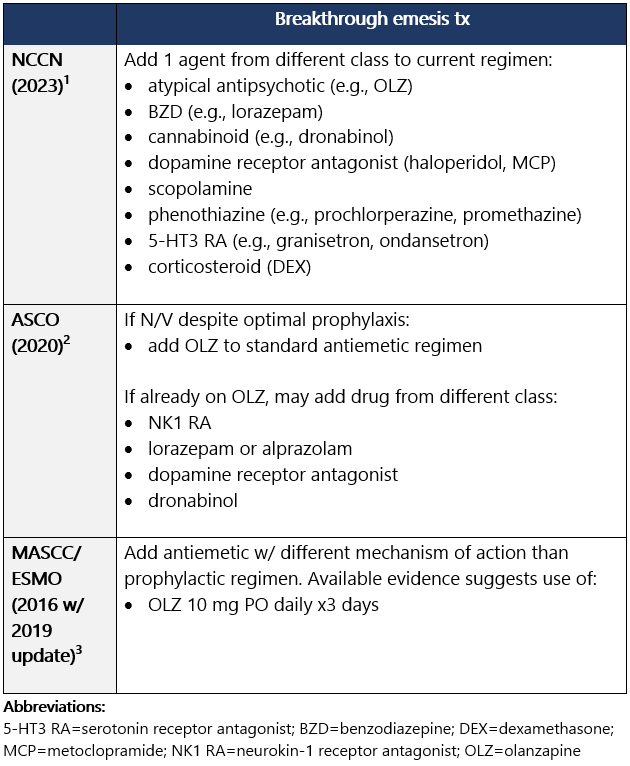
Guideline recommendations for breakthrough N/V: General principles for managing breakthrough emesis: - Antiemetics are most effective when used prophylactically: Use maximally effective antiemetics as 1st-line tx rather than withholding them for later use.3
- If breakthrough N/V, re-eval emetic risk, dz status, concurrent illnesses, and meds; ensure optimal antiemetic regimen is being administered.2
- Give additional agent from different drug class; some pts may require several agents w/ different mechanisms of action.1
- Strongly consider around-the-clock admin vs. prn dosing.1
- PO route not likely feasible, consider rectal or IV.1
- Multiple agents, perhaps in alternating schedules or routes, may be needed.1
- Ensure adequate fluids, check electrolytes.1
- Consider non-anticancer tx–related reasons for emesis: brain mets, electrolyte abnormalities, tumor infiltration of bowel, etc.1
- Prior to next cycle, reassess day-1 and post-anticancer tx antiemetic regimen.1
- Consider antacid tx (H2 blocker or PPI) if pt has dyspepsia.1
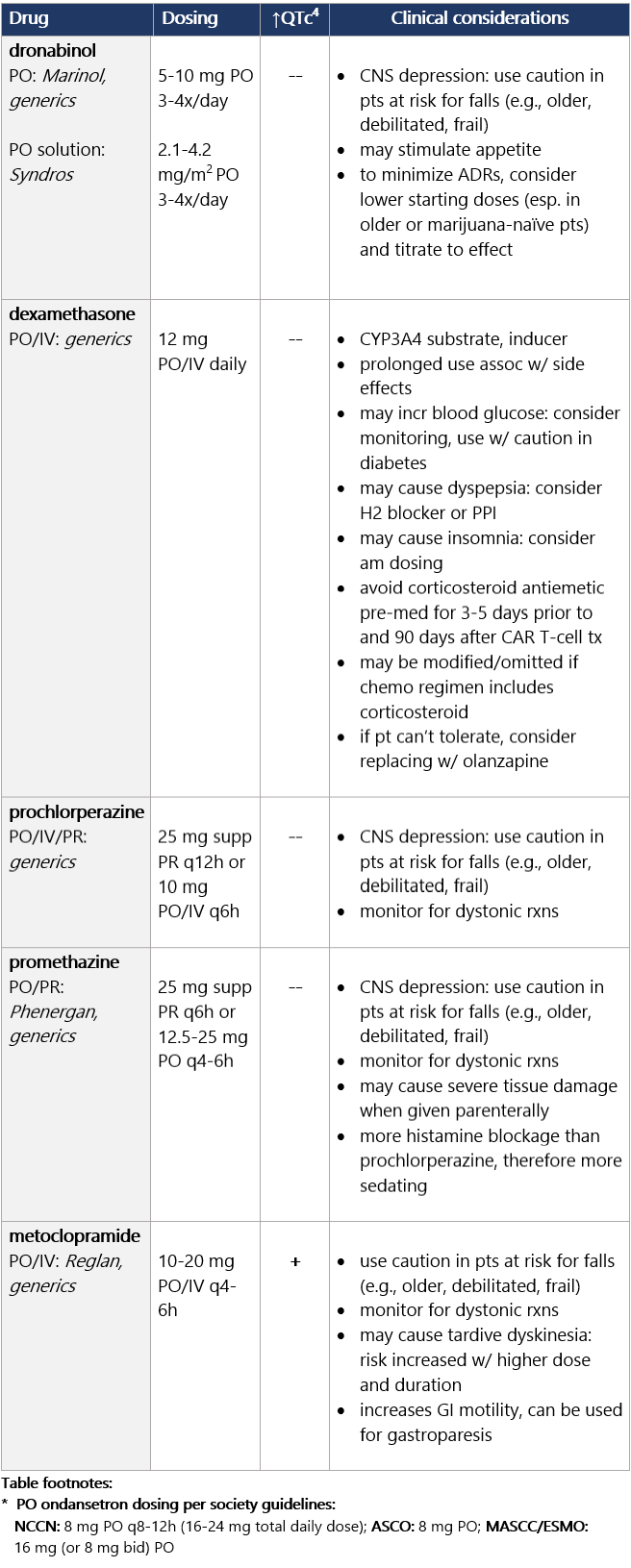
Options for breakthrough N/V Options for breakthrough N/V (continued) View epocrates drug info: Footnotes 1 NCCN 2023. National Comprehensive Cancer Network. Antiemesis (Version 1.2024). December 13, 2023. Accessed December 20, 2023
2 ASCO 2020. Hesketh PJ, et al. Antiemetics: ASCO Guideline Update. J Clin Oncol. 2020. 2020. Aug 20;38(24):2782-2797. Accessed October 19, 2020
• For adults, addition of a checkpoint inhibitor (CPI) to chemo doesn’t change the guideline rec for antiemetic regimen based on emetogenicity of chemo agents; CPIs are minimally emetogenic and don’t require a prophylactic antiemetic.
3 MASCC/ESMO 2016/2019. Roila F, et al. 2016 MASCC and ESMO Guideline Update for the Prevention of Chemotherapy- and Radiotherapy-Induced Nausea and Vomiting and of Nausea and Vomiting in Advanced Cancer Patients. Ann Oncol. 2016. Sep;27(suppl 5):v119-v133. PDF
Aapro M, et al. MASCC/ESMO Antiemetic Guideline 2016 With Updates in 2019. June 2019. Multinational Association of Supportive Care in Cancer.
4 Barni S, et al. Cardiotoxicity of Antiemetic Drugs in Oncology: An Overview of the Current State of the Art. Crit Rev Oncol Hematol. 2016. June;102:125-34. PubMed® abstract
5 IE: insufficient evidence. QT prolongation data for PO/IV/SC granisetron are limited and inconsistent.
6 Granisetron transdermal doesn’t alter QT interval.
7 Ondansetron: dose-dependent QT change.
|