-
To prevent/delay progression to T2DM and maintain QOL: Use lifestyle modifications, consider metformin, monitor regularly; discuss DM technology-assisted tools for self-mgmt/support - Refer pts w/ IGT, IFG,1 or A1C 5.7%-6.4%2 to intensive diet/physical activity behavioral counseling program targeting 7% body wt loss & increasing mod physical activity (eg, brisk walking) to at least 150min/wk [A]
- Metformin3,4 to prevent T2DM has the strongest evidence and long-term safety profile: consider if BMI ≥35 kg/m2, age <25-59 yo, FPG ≥110 mg/dL, A1C ≥6.0%, or prior hx gestational DM [A]
- Variety of eating patterns acceptable for pts w/ prediabetes [B], incl Mediterranean and low-cal, low-fat diets
- DM self-mgmt education/support programs may be appropriate in order to develop/maintain behaviors that can prevent/delay development of T2DM [B]
- Monitor at least annually [E] for the development of T2DM; modify freq based on individual risk/benefit assessment (using informal risk assessment or an assessment tool); see Diabetes Risk Score (Type 2) in epocrates Calculators
- Screen for & treat modifiable CV risks5 [B]
- Based on pt preference, consider DM technology-assisted tools to help prevent/delay onset of T2DM [B]
- Consider referral to RDN for individualized medical nutrition tx
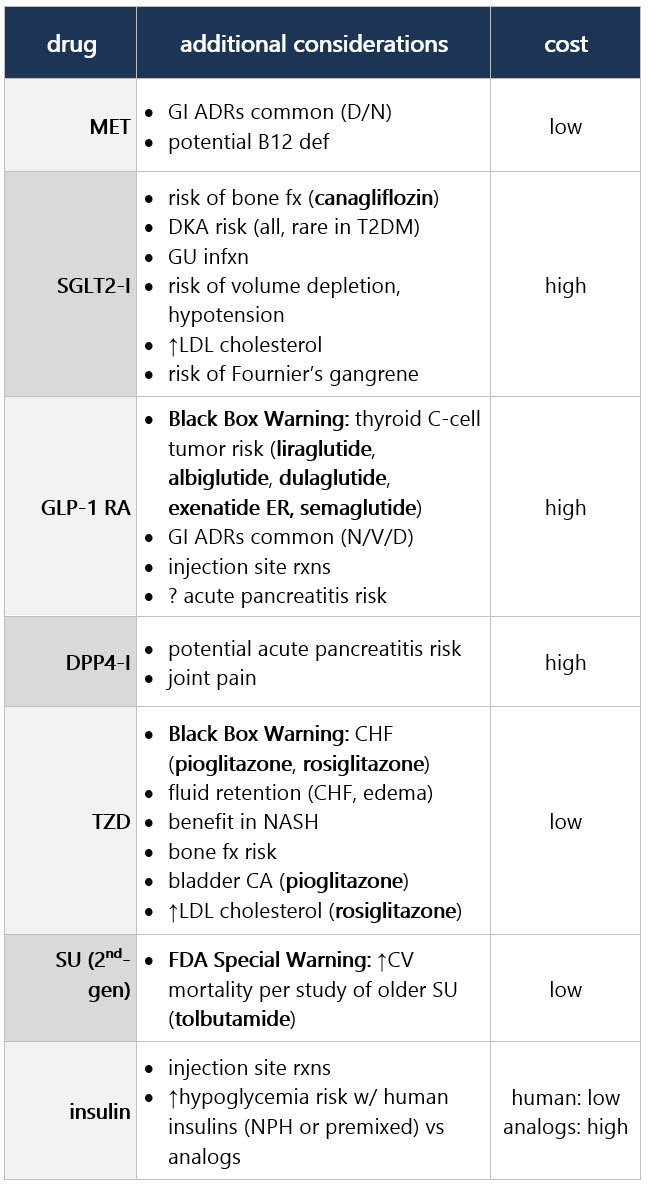
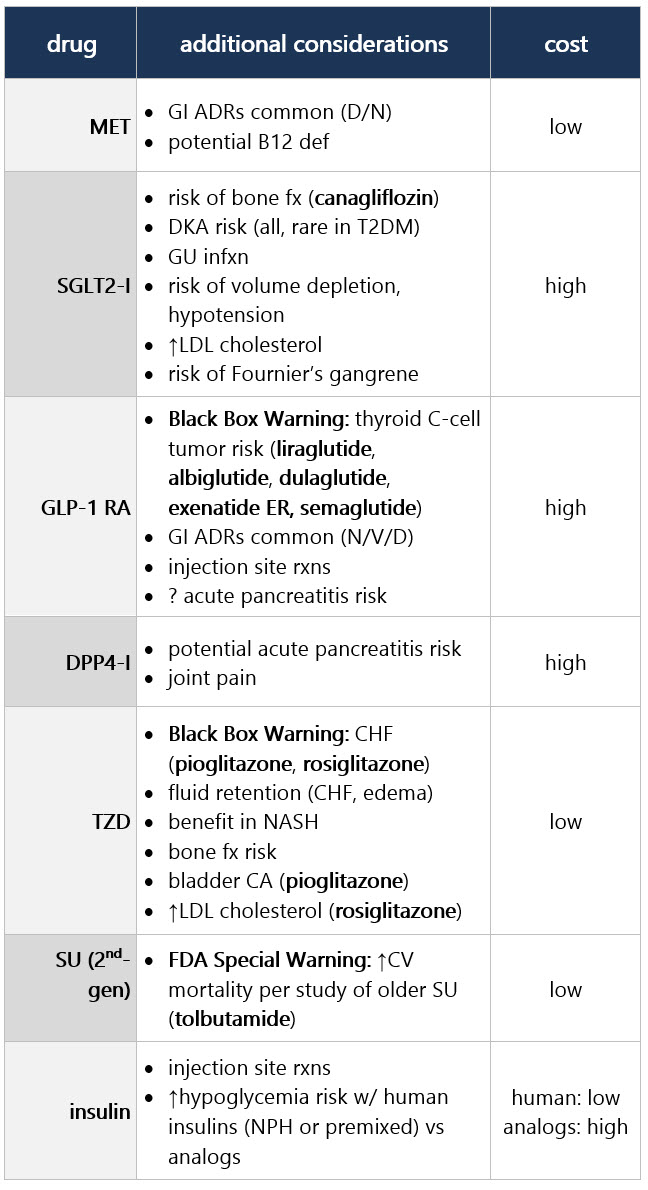
- Obesity mgmt/wt loss. No clear evidence that dietary supplements (eg, herbs & botanicals, high-dose vitamins & minerals, amino acids, enzymes, antioxidants, etc) are effective for obesity mgmt/wt loss [A].6 Supplementation of certain vitamins & minerals (eg, iron, vit B12, vit D) may be indicated in pts w/ documented deficiency; protein supplements may also be appropriate as adjuncts to medically supervised wt loss regimens.
Footnotes 1 IGT (impaired glucose tolerance)=75-g OGTT 2-h PG 140-199 mg/dL; IFG (impaired fasting glucose)=FPG 100-125 mg/dL.
2 Using A1C to screen for prediabetes may be affected by presence of certain hemoglobinopathies or conditions affecting RBC turnover.
3 In addition to metformin, α-glucosidase inhibitors, GLP-1 RAs, TZDs, and several agents approved for wt loss have been shown to decrease incident T2DM to various degrees in pts w/ prediabetes.
4 Due to the assoc of long-term metformin use w/ biochemical vit B12 def, consider periodic measurement of vit B12 levels, esp in pts w/ anemia or peripheral neuropathy [B].
5 Prediabetes is assoc w/ heightened CV risk; people w/ prediabetes often have other CV risk factors such as HTN and dyslipidemia.
6 Several large systematic reviews show that most trials evaluating dietary supplements for wt loss are of low quality & @ high risk for bias. High-quality published studies show little or no wt loss benefits.
-
Diabetes already diagnosed, nonpregnant
Use pt-centered approach to guide initial med choice; meds other than metformin may be appropriate initial agent in pts w/ ASCVD, HF, CKD and in those wishing to maintain/lose wt; don’t delay tx intensification if not meeting goals; GLP-1 RA preferred to insulin in most pts; reassess and modify tx every 3-6mo as needed - 1st-line tx depends on comorbidities, pt-centered treatment factors/mgmt needs - generally includes metformin1 and comprehensive lifestyle modification, but other agents may also be appropriate initial choice [A]
- If pt has established atherosclerotic CV dz or indicators of high risk, established kidney dz, or HF, an SGLT2-I or GLP-1 RA w/ demonstrated CV/renal dz benefit is recommended as part of tx regimen independent of A1C [A]
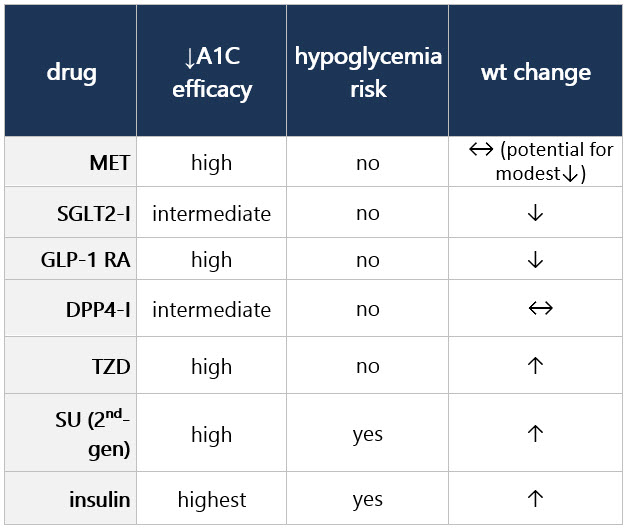
- If pt wishes to minimize wt gain or promote wt loss, a GLP-1 RA w/ good efficacy for wt loss or SGLT2-I is preferred
- If evidence of catabolism (wt loss, ketosis), sx of hyperglycemia (ie, polyuria, polydipsia), and/or A1C ≥10% or blood glucose ≥300 mg/dL: Consider early initiation of insulin [E]
- Consider early combo tx at tx initiation in some pts to extend time to tx failure [A]
- In pts who need greater glucose lowering than can be obtained w/ oral agents, GLP-1 RAs are preferred to insulin when possible [B]
- If insulin is used, combo tx w/ GLP-1 RA is recommended to improve efficacy and durability of tx effect [A]
- Reassess/individualize glycemic targets over time based on modifiable/non-modifiable pt/dz features2,3 [E]
- Reasonable glycemic goal in many nonpregnant adults is A1C <7% [A] w/o significant hypoglycemia.4 Based on clinician judgment/pt preference, lower goal (eg, <6.5%) may be acceptable if can safely be achieved w/o significant hypoglycemia/other adverse effects [C]. Less stringent goals (eg, <8%) may be appropriate in those w/ hx of severe hypoglycemia, limited life expectancy, advanced micro/macro-vascular dz, extensive comorbidities, or long-standing DM despite education, glucose monitoring, and use of meds incl insulin [B]
- Assess glycemic status (A1C or other glycemic measurement)5 at least twice yearly if pt meeting goals/stable glycemic control; quarterly if not meeting goals or tx has changed; point-of-care testing allows for timelier tx changes [E]
- Review occurrence & risk of hypoglycemia at every encounter & investigate as indicated [C]. Prescribe glucagon for pts at increased risk of hypoglycemia [E]. Trigger hypoglycemia avoidance education w/ hypoglycemia unawareness or ≥1 level 3 hypoglycemic episodes [E]. Consider raising glycemic targets for several wks or longer w/ hypoglycemia unawareness, one level 3 hypoglycemic event, or pattern of unexplained level 2 hypoglycemic events [A]
- Don’t delay intensified tx for pts not meeting tx goals [B]
- If overwt/obese: Achieve ≥5% wt loss via multifaceted approach, incl: diet, physical activity & behavioral therapy [B]. Wt loss meds or bariatric surgery may be appropriate for some obese pts [A]. No clear evidence that dietary supplements (eg, herbs & botanicals, high-dose vitamins & minerals, amino acids, enzymes, antioxidants, etc) are effective for obesity mgmt/wt loss [A].6 Supplementation of certain vitamins & minerals (eg, iron, vit B12, vit D) may be indicated in pts w/ documented deficiency; protein supplements may also be appropriate as adjuncts to medically supervised wt loss regimens
- Micronutrients/herbal supplements: Not recommended for glycemic control [C]7 in pts w/o underlying deficiencies
- Re-eval med regimen every 3-6mo and adjust prn to account for pt factors and regimen complexity [E]
Footnotes 1 Due to the assoc of long-term metformin use w/ biochemical vit B12 def, consider periodic measurement of vit B12 levels, esp in pts w/ anemia or peripheral neuropathy [B].
2 Individualize glycemic targets based on risks potentially assoc w/ hypoglycemia and other drug adverse effects, dz duration, life expectancy, comorbidities, established vascular complications, pt preference, and resources and support system [E].
3 Treatment considerations in adults ≥65 yo:
Consider assessment of medical, psychological, functional (self-mgmt abilities), and social domains to provide framework to determine targets/tx approaches [B]. Screen for polypharmacy, cognitive impairment, depression, urinary incontinence, falls, pain in older adults as they may affect self-mgmt and diminish QoL [B]. Prefer med classes w/ low risk of hypoglycemia [B]; avoid overtx [B]; deintensify/simplify complex regimens to lower hypoglycemia risk, if doable w/in individualized A1C target [B].
A1C targets:
• Healthy older adults (few coexisting chronic illnesses, intact cognitive fxn, & functional status) should have lower glycemic goals (such as A1C <7.0%-7.5%) [C].
• Complex/intermediate older adults w/ multiple coexisting chronic illnesses, cognitive impairment, or functional dependence should have less stringent glycemic goals (such as A1C <8.0%) [C].
• Community-dwelling pts (skilled nursing facility or short-term rehabilitation): Avoid reliance on A1C; glucose target: 100-200 mg/dL.
• Very complex/poor health: Avoid reliance on A1C. Avoid hypoglycemia & symptomatic hyperglycemia.
• At end of life: Avoid hypoglycemia & symptomatic hyperglycemia
4 If using ambulatory glucose profile/glucose management indicator to assess glycemia, a parallel goal is time in target range (70-180 mg/dL).
5 Self-monitored blood glucose and continuous glucose monitoring provide information about glycemic variability and hypoglycemic episodes not reflected in A1C measurements.
6 Several large systematic reviews show that most trials evaluating dietary supplements for wt loss are of low quality & @ high risk for bias. High-quality published studies show little or no wt loss benefits.
7 There’s no clear evidence that dietary supplementation w/ vitamins, minerals (such as chromium and vit D), herbs, or spices (such as cinnamon, curcumin, or aloe vera) can improve outcomes in pts w/ DM who don’t have underlying deficiencies:
• Routine supplementation w/ antioxidants (such as vit E, vit C, and carotene) not advised d/t lack of evidence of efficacy & concern related to long-term safety.
• More research needed to define pt characteristics & clinical factors where vit D supplementation may benefit.
• However, in special populations (incl pregnant/lactating women, older adults, vegetarians, and pts following very low-calorie or low-carbohydrate diets) a multivitamin may be necessary.
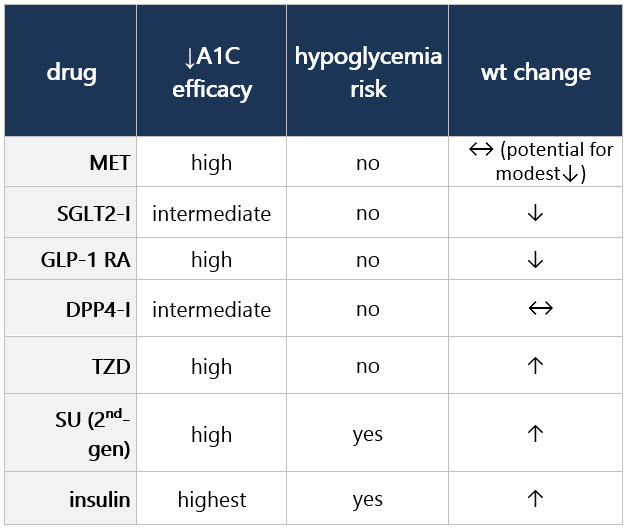
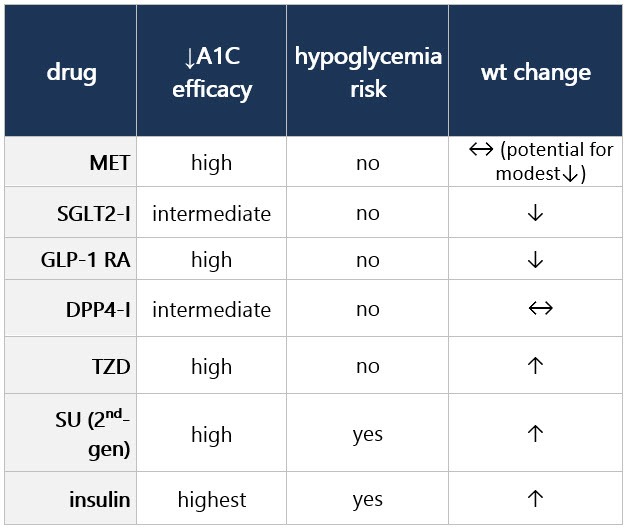
Tx-experienced (on ≥1 drugs), not at A1C target On mono-tx, A1C not at target No ASCVD (or high risk of) HF or CKD Consider pt factors, including age, when choosing drug add-ons - Base drug choice on factors such as hypoglycemia risk, wt gain potential, and cost
- In pts ≥65 yo: Consider coexisting conditions/cognition/functional status w/ care to avoid symptomatic hyperglycemia and minimize risk of acute hyperglycemia complications [B]1
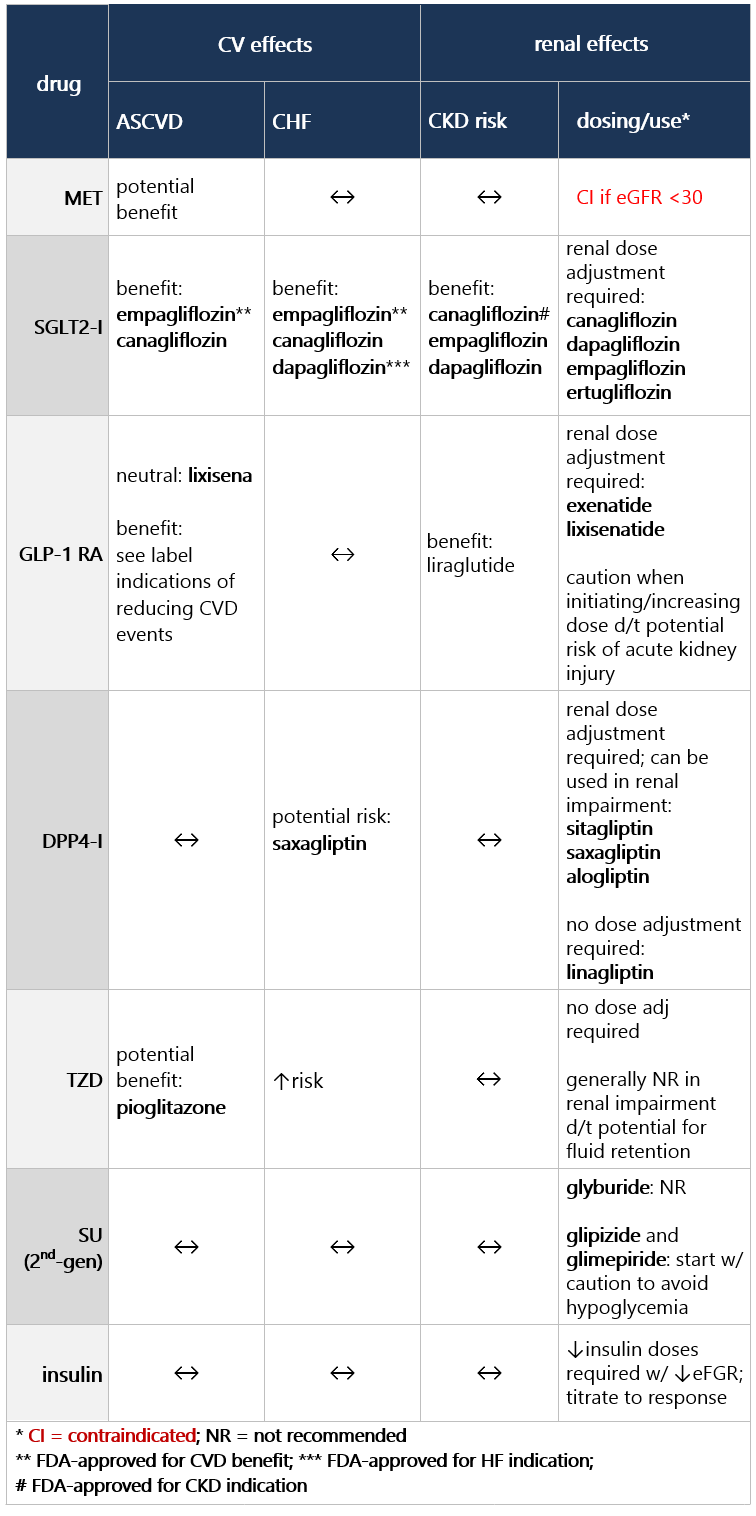
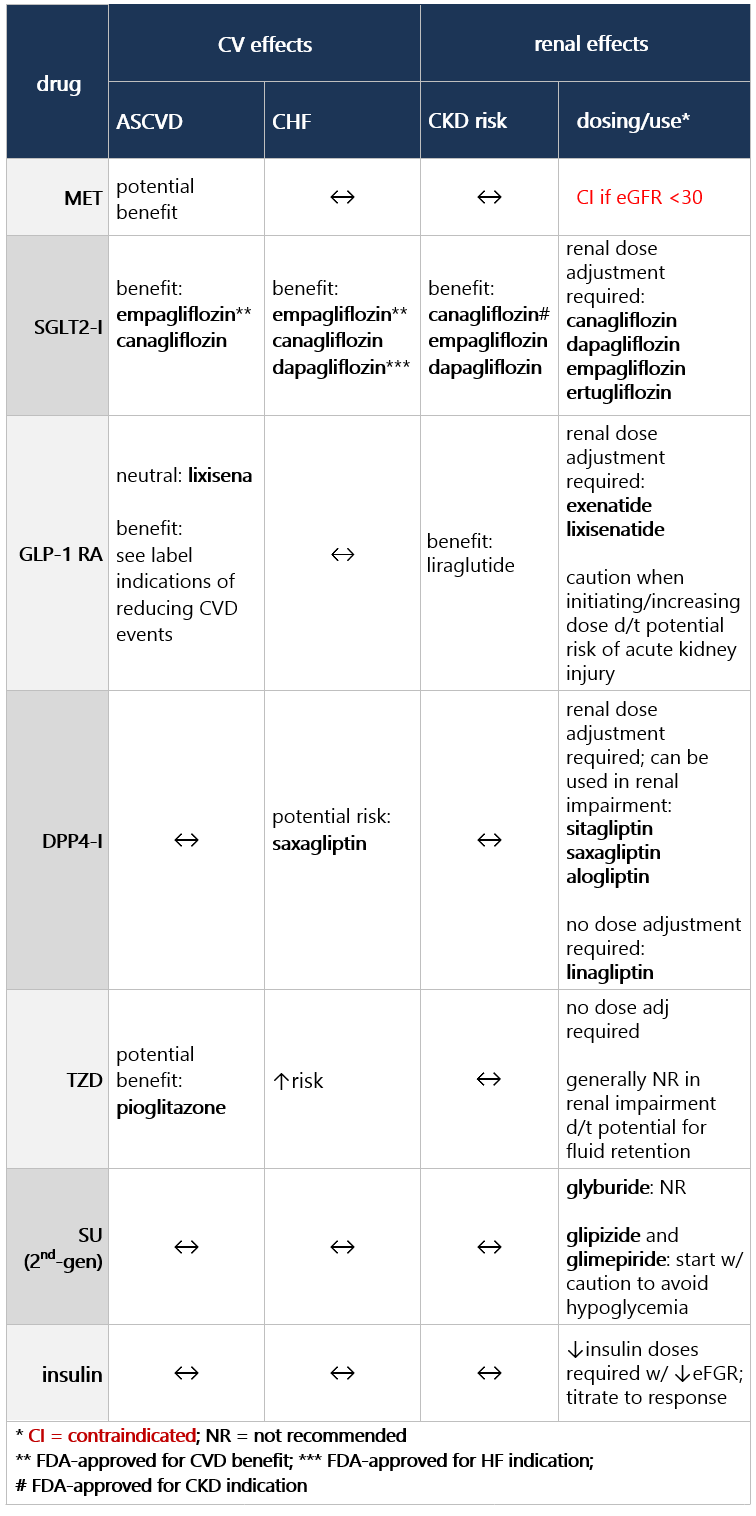
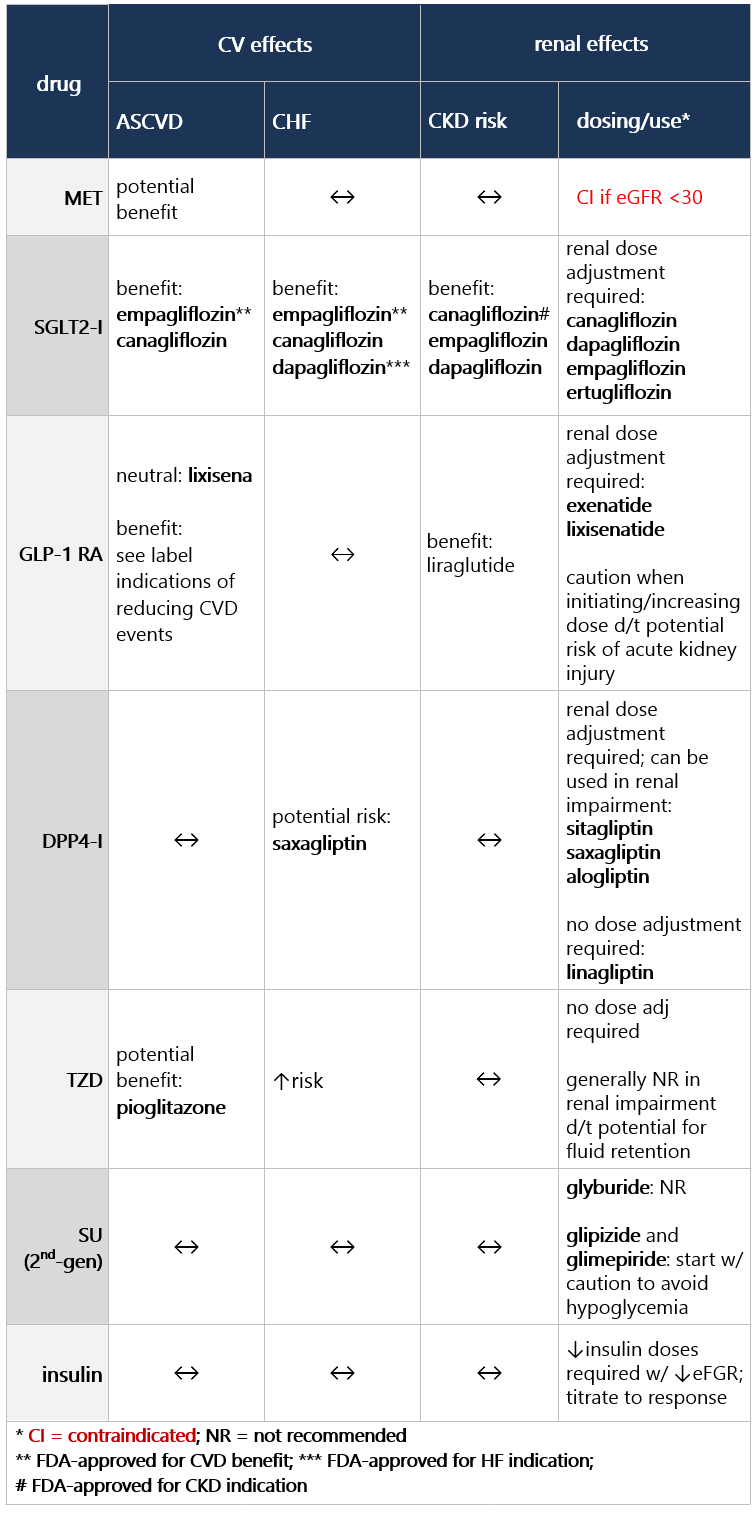
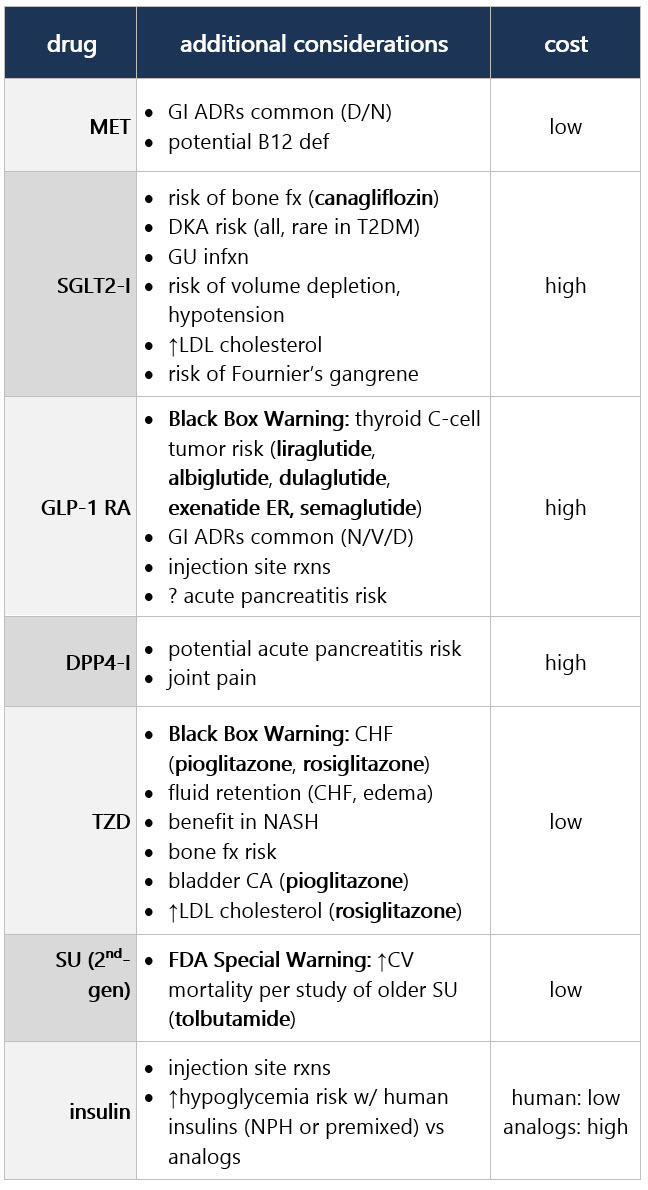
If compelling need to minimize hypoglycemia (eg, older age):1 If compelling need to minimize wt gain or promote wt loss: If cost/access is a major issue: Efficacy, hypoglycemia risk,1 and wt concerns to consider when selecting antihyperglycemic tx: CV and renal effects to consider when selecting antihyperglycemic tx: Additional considerations when selecting antihyperglycemic tx: Footnotes 1 Treatment considerations in adults ≥65 yo:
Consider assessment of medical, psychological, functional (self-mgmt abilities), and social domains to provide framework to determine targets/tx approaches [B]. Screen for polypharmacy, cognitive impairment, depression, urinary incontinence, falls, pain in older adults as they may affect self-mgmt and diminish QoL [B]. Prefer med classes w/ low risk of hypoglycemia [B]; avoid overtx [B]; deintensify/simplify complex regimens to lower hypoglycemia risk, if doable w/in individualized A1C target [B].
A1C targets:
• Healthy older adults (few coexisting chronic illnesses, intact cognitive fxn, & functional status should have lower glycemic goals (such as A1C <7.0%-7.5%) [C].
• Complex/intermediate older adults w/ multiple coexisting chronic illnesses, cognitive impairment, or functional dependence should have less stringent glycemic goals (such as A1C <8.0%) [C].
• Community-dwelling pts (skilled nursing facility or short-term rehabilitation): Avoid reliance on A1C; glucose target: 100-200 mg/dL.
• Very complex/poor health: Avoid reliance on A1C. Avoid hypoglycemia & symptomatic hyperglycemia.
• At end of life: Avoid hypoglycemia & symptomatic hyperglycemia.
2 SGLT-I vary by region and individual agent as far as indicated level of eGFR for initiation and continued use.
3 Consider country- and region-specific drug costs. ASCVD (established or high risk) If established ASCVD or indicators of high ASCVD risk,1 incorporate either a GLP-1 RA w/ proven CVD benefit or a SGLT2-I w/ proven CVD benefit (if eGFR adequate) [A]: - For pts on a GLP-1 RA, consider adding SGLT2-I w/ proven CVD benefit and vice versa2
- TZD also an option (low dose may be better tolerated, but less well studied for CVD effects), but ↑risk of HF
If further tx intensification needed (A1C still above target) or pt now unable to tolerate GLP-1 RA or SGLT2-I, consider tx intensification based on co-morbidities, pt-centered tx factors, & mgmt needs; choose agents demonstrating CV safety/neutrality: - metformin (potential CV benefit)
- DPP4-I (but potential ↑risk of HF w/ saxagliptin)
- SU (choose later gen, w/ lower hypoglycemia risk)
- insulin3
If pt ≥65 yo: Consider coexisting conditions/cognition/functional status w/ care to avoid symptomatic hyperglycemia and minimize risk of acute hyperglycemia complications [B]4 Efficacy, hypoglycemia risk,4 and wt concerns to consider when selecting antihyperglycemic tx: CV and renal effects to consider when selecting antihyperglycemic tx: Additional considerations when selecting antihyperglycemic tx: Footnotes 1 Indicators of high ASCVD risk: Age ≥55 yo w/ coronary, carotid, or lower extremity artery stenosis >50%, or LVH.
2 Proven CVD benefit means it has label indication of reducing CVD events.
3 Basal insulin alone is the most convenient initial insulin regimen. Clinical signals that should prompt evaluation for overbasalization w/ insulin tx include: basal dose >0.5 units/kg, high bedtime-morning or postprandial glucose differential, hypoglycemia (aware or unaware), and high variability.
4 Treatment considerations in adults ≥65 yo:
Consider assessment of medical, psychological, functional (self-mgmt abilities), and social domains to provide framework to determine targets/tx approaches [B]. Screen for polypharmacy, cognitive impairment, depression, urinary incontinence, falls, pain in older adults as they may affect self-mgmt and diminish QoL [B]. Prefer med classes w/ low risk of hypoglycemia [B]; avoid overtx [B]; deintensify/simplify complex regimens to lower hypoglycemia risk, if doable w/in individualized HbA1C target [B].
A1C targets:
• Healthy older adults (few coexisting chronic illnesses, intact cognitive fxn, & functional status should have lower glycemic goals (such as A1C <7.0%-7.5%) [C].
• Complex/intermediate older adults w/ multiple coexisting chronic illnesses, cognitive impairment, or functional dependence should have less stringent glycemic goals (such as A1C <8.0%) [C].
• Community-dwelling pts (skilled nursing facility or short-term rehabilitation): Avoid reliance on A1C; glucose target: 100-200 mg/dL.
• Very complex/poor health: Avoid reliance on A1C. Avoid hypoglycemia & symptomatic hyperglycemia.
• At end of life: Avoid hypoglycemia & symptomatic hyperglycemia.
If HF (particularly LVEF ≤40%): - Use an SGLT2-I w/ evidence of reducing HF in CV outcome trials1 [A]
- Can continue metformin in stable HF (as long as eGFR >30), but avoid if unstable or hospitalized HF [B]
- Avoid TZD in setting of HF; also potential ↑risk of worsening HF w/ saxagliptin
If CKD (specifically eGFR <60): - For pts w/ albuminuria (UACR ≥300 mg/g) and eGFR ≥25: Preferably use SGLT2-I w/ evidence of reducing CKD progression (in either primary CKD or CV outcomes trials); if SGLT2-I contraindicated (ie, eGFR <25) or not tolerated: use GLP1-RA w/ proven CVD benefit1,2
- If CKD w/o albuminuria, use either GLP1-RA w/ proven CVD benefit or or SGLT2-I w/ proven CVD benefit1,2
If further tx intensification needed (A1C still above target) or pt unable to tolerate GLP-1 RA or SGLT2-I, consider tx intensification based on co-morbidities, pt-centered tx factors, & mgmt needs; choose agents demonstrating CV safety/neutrality: - For pts on SGLT2-I, consider adding GLP-1 RA w/ proven CVD benefit and vice versa2
- metformin (potential CV benefit)
- DPP4-I (but potential ↑risk of HF w/ saxagliptin)
- SU (choose later gen, w/ lower hypoglycemia risk)
- insulin3
If pt ≥65 yo: Consider coexisting conditions/cognition/functional status w/ care to avoid symptomatic hyperglycemia and minimize risk of acute hyperglycemia complications [B]4 Efficacy, hypoglycemia risk,4 and wt concerns to consider when selecting antihyperglycemic tx: CV and renal effects to consider when selecting antihyperglycemic tx: Additional considerations when selecting antihyperglycemic tx: Footnotes 1 Evidence of benefit for HF: empagliflozin, canagliflozin, dapagliflozin, ertugliflozin
Evidence of benefit for CKD: empagliflozin, canagliflozin, dapagliflozin, liraglutide, semaglutide, dulaglutide
2 Proven CVD benefit means it has label indication of reducing CVD events.
3 Basal insulin alone is the most convenient initial insulin regimen. Clinical signals that should prompt evaluation for overbasalization w/ insulin tx include: basal dose >0.5 IU/kg, high bedtime-morning or postprandial glucose differential, hypoglycemia (aware or unaware), and high variability. Lower insulin doses req’d with decrease in eGFR.
4 Treatment considerations in adults ≥65 yo:
Consider assessment of medical, psychological, functional (self-mgmt abilities), and social domains to provide framework to determine targets/tx approaches [B]. Screen for polypharmacy, cognitive impairment, depression, urinary incontinence, falls, pain in older adults as they may affect self-mgmt and diminish QoL [B]. Prefer med classes w/ low risk of hypoglycemia [B]; avoid overtx [B]; deintensify/simplify complex regimens to lower hypoglycemia risk, if doable w/in individualized A1C target [B].
A1C targets:
• Healthy older adults (few coexisting chronic illnesses, intact cognitive fxn, and functional status should have lower glycemic goals (such as A1C <7.0%-7.5%) [C].
• Complex/intermediate older adults w/ multiple coexisting chronic illnesses, mild-to-mod cognitive impairment, or functional dependence should have less stringent glycemic goals (such as A1C <8.0%) [C].
• Community-dwelling pts (skilled nursing facility or short-term rehabilitation): Avoid reliance on A1C; glucose target: 100-200 mg/dL.
• Very complex/poor health: Avoid reliance on A1C. Avoid hypoglycemia & symptomatic hyperglycemia.
• At end of life: Avoid hypoglycemia and symptomatic hyperglycemia. On dual or triple tx, A1C not at target If injectable tx needed to reduce A1C: - Consider GLP-1 RA1 in most pts prior to insulin [B]: Initiate appropriate start dose (varies by agent w/in class) and gradually titrate to maintenance (varies by agent)
- If already on GLP-1 RA, GLP-1 RA not appropriate, or insulin preferred, see Add basal insulin below
- Consider insulin as the 1st injectable if: evidence of ongoing catabolism; sx of hyperglycemia present; A1C >10% or blood glucose levels are very high (≥300 mg/dL; or dx of T1DM is a possibility)
- If insulin is used, combo tx w/ GLP-1 RA is recommended to improve efficacy and durability of tx effect [A]
If A1C still above target, add basal insulin2 (basal analog or bedtime NPH insulin) - Initiation: Start 10 units per day OR 0.1-0.2 units/kg per day
- Titration: Set FPG target; choose evidence-based titration algorithm (eg, increase 2 units q3 days to reach FPG target w/o hypoglycemia); for hypoglycemia, determine cause; if no clear reason: Lower dose by 10%-20%
If A1C still above target despite adequately titrated basal analog or bedtime NPH3 OR once-daily basal dose >0.5 units/kg OR FPG at target - If on bedtime NPH: Consider converting to twice-daily NPH regimen.4 Then, if still above A1C target: Add prandial insulin as below
- Otherwise, add prandial insulin.5 If adding prandial insulin to NPH: Consider a self-mixed or pre-mixed insulin regimen to decrease # of injections
If A1C still above target, consider 1 of 3 options: - Stepwise additional injections of prandial insulin (ie, 2, then 3 injections); then proceed to full basal-bolus regimen (ie, basal insulin and prandial insulin w/ each meal)
- Self-mixed/split insulin regimen (can adjust NPH and short/rapid-acting insulins separately)6
- Twice-daily premix insulin regimen7
Footnotes 1 When selecting GLP-1 RA, consider: pt preference, A1C lowering, wt lowering effect, or freq of injection. If CVD: Consider GLP-1 RA w/ proven CVD benefit.
2 For pts on GLP-1 RA and basal insulin combo: Consider fixed-ratio combo product (iDegLira or iGlarLixi).
3 Consider switch from evening NPH to basal analog if pt develops hypoglycemia and/or frequently forgets to administer NPH in the evening and would be better managed w/ morning dose of long-acting basal insulin.
4 Conversion to BID NPH insulin based on individual needs and current glycemic control. One possible approach:
• Initiation: Total dose = 80% of current bedtime NPH dose; 2/3 given in AM, 1/3 at bedtime.
• Titration:based on individual needs.
5 Prandial insulin: Usually 1 dose w/ largest meal or meal w/ greatest PPG excursion; prandial insulin can be dosed individually or mixed w/ NPH as appropriate.
• Initiation: 4 units/day or 10% of basal insulin dose; if A1C <8%: Consider lowering basal dose by 4 units/day or 10% of basal dose.
• Titration: Increase dose by 1-2 units or 10%-15% twice weekly; for hypoglycemia, determine cause; if no clear reason, lower corresponding dose by 10%-20%.
6 Self-mixed/split insulin regimen:
• Initiation: Total NPH dose = 80% of current NPH dose; 2/3 given before breakfast; 1/3 given before dinner; add 4 units short/rapid-acting insulin to each injection or 10% of reduced NPH dose.
• Titration: Titrate each component of regimen based on individual needs.
7 Twice-daily premix insulin regimen:
• Initiation: Usually unit per unit at same total insulin dose, but may require adjustment to individual needs.
• Titration: Titrate based on individual needs.
|